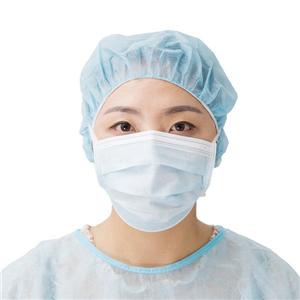
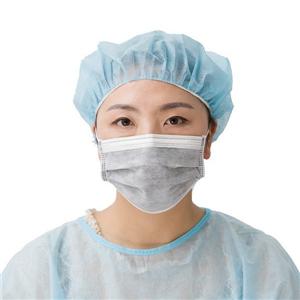
CE/MDR-EU Quality Assurance Certificate

For medical product, MDR is the complusory document to access to EU market.
Pursuant to Regulation (EU) 2017/745 on Medical Devices, Annex XI Part A.
Get the latest price? We'll respond as soon as possible(within 12 hours)
more products
Products
Featured Products
Contact Details